For years, hair transplant (HT) surgeons have debated the best medium for short-term storage of hair-bearing grafts during the period prior to placement in the recipient area, and the temperature at which the medium should be maintained. Normal saline (NS), lactated Ringer’s solution, and others have been utilized, at room temperature and at varying degrees of coldness. The importance of this may be reflected in graft survival and yield, in post-operative effluvium, and in graft retention. However, it remains unclear as to the most advantageous protocol for ex vivo storage of hair-bearing grafts.
It is well known that tissues continue to consume energy in metabolic processes, as well as to create waste products when excised from living organisms and placed in aqueous solutions. This, of course, may quickly deplete energy stores and create increasing levels of free radicals, along with a decrease in pH as an acidic environment develops. Prior studies have shown that cells become metabolically quite inactive at temperatures of 4 degrees Celsius or lower. It has also been shown that buffered saline solution (BSS) maintains a constant pH over time (with or without tissue, up to 5 hours), as opposed to plain normal saline, which reveals a drop in pH quickly and continues to decline as a function of time independent of temperature or the presence of graft tissue.
There are a number of reasons to consider avoiding the use of saline as a storage solution for hair-bearing grafts in hair transplant surgery. One is the drop in pH that occurs over time with room temperature saline. This may have a deleterious effect on graft survival and eventual yield; the lack of metabolic slow-down observed with room temperature solutions is another consideration. Although cold storage solutions markedly decrease the metabolic activity of tissues, the cold saline solution causes paralysis of sodium-potassium active transport channels; the end result of this phenomenon is a net influx of water and resultant cellular edema, or swelling. If unchecked, this may lead to lysis or bursting of the cell.
To elaborate on the above, let us consider the membrane pump which maintains the intracellular concentration of potassium many times above that in the extracellular space (outside the cell). It also maintains the concentration of sodium many times lower than that outside. All other ions are sequestered in a similar manner in order to maintain electrical and osmotic balance. When the ion pump mechanism is perturbed by, for example, decreased temperature, the electrical and osmotic gradients are disrupted. As a result, water enters the cell, attracted by solutes and colloidal materials; then, electrically charged ions follow in an attempt to normalize the trans-membrane potential. The resultant hypertonic state leads to even more water passively crossing into the cell, and cellular edema exists.
These phenomena discussed above have to lead to the development of various hypothermic cell preservation solutions. These are designed to be used exclusively in a cold environment. Studies comparing human hair follicles stored in saline and in a cold tissue solution (Hypothermosol or HTS) showed a significant improvement in viability of the Hypothermosol tissues, both in terms of metabolic activity during recovery, and in terms of cell membrane integrity. Specialized cold tissue storage media such as HTS are designed to achieve several goals in which saline-type solutions cannot; that is, to minimize swelling, maintain ionic balance across cell membranes, prevent acidosis, prevent free radical formation, or quench free radicals, and to provide high-energy substrates upon reperfusion. It is well established that the reperfusion period, during which ischemic tissues have blood flow restored, is most critical for the viability and survival of grafted tissues.
We have developed a technique that utilizes circulating coolant to maintain our hair-bearing grafts in cold HTS during a time out of the body. Preliminary observation suggests that yields post-hair transplants may be superior using this technique. The set-up is relatively simple. A pump circulates cold water via inflow and outflow conduits. These conduits communicate by way of a “capillary bed” arrangement of sorts; it is above this “bed” that the graft containers are placed. Pre-chilled Hypothermosol is placed in the containers (eg, Petri dishes) and monitored to the desired temperature, which in our facility is 2 to 4 degrees Celsius. The temperature is monitored continuously with a laser thermometer directed into the solution.
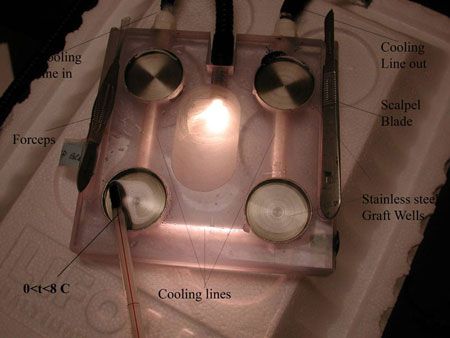
We have learned that the optimal temperature for maintaining tissues is about 2 to 4 degrees Celsius. This is easily maintained with our apparatus (as our continual monitoring reveals), and the inclusion of Hypothermosol as the medium of choice. Further studies will be needed to evaluate the clinical efficacy of this promising technique.